Canadian-Led lmmunotherapies in Cancer: CLIC-1901 for the Treatment of Patients with Relapsed/Refractory CD19 Positive Hematologic Malignancies
Key Information
Who may qualify?
- Participants must have relapsed or refractory CD19+ disease
- All eligible participants must have documentation of CD19 tumour expression demonstrated in tissue biopsy, bone marrow or peripheral blood within the 3 months prior to study screening.
- Adequate organ function
- Participant age: 18 to 75 years.
- For full inclusion criteria, please click the link at the bottom of the page
Recruitment status
- Active; recruiting
Key words
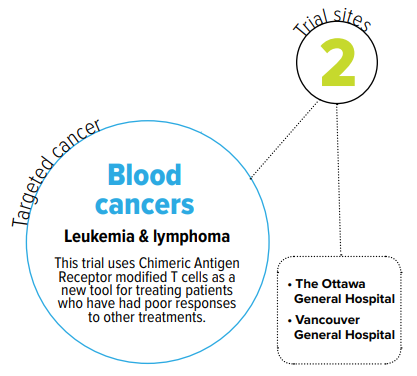
- CAR-T, blood cancer, leukemia, lymphoma, Canadian, CLIC-1901, t cells
About the Trial
Patients with certian types of blood cancer that don’t respond to standard therapies have a particularly poor chance of survival. Chimeric Antigen Receptor modified T cells (CAR-Ts) are a powerful new tool for treating these patients. CAR-T cells are made by isolating a sample of a patient’s T lymphocytes (a type of white blood cell) from their blood, genetically modifying and activating the cells in the lab, and then re-administering them to the same patient, allowing for a patient’s immune cells to be targeted against their tumour.
The evidence to support for the use of CAR-T cells in certain leukemias and lymphomas has been phenomenal in some cases, with durable responses, suggesting that patients who respond are cured of their underlying malignancy. Because CAR-T therapy is very personalized (it requires genetically engineering the patient’s own cells), there is considerable infrastructure and expertise required to manufacture and deliver this treatment safely and successfully.
This is the first clinical trial of Canadian-made CAR-T cells. The goal of this trial is to determine the feasibility, safety, and efficacy of administering CLIC-1901 cells manufactured in a manner compatible with a distributed point of care model.
(https://bit.ly/35JvvVq)
Clinical Trial #: NCT03765177

