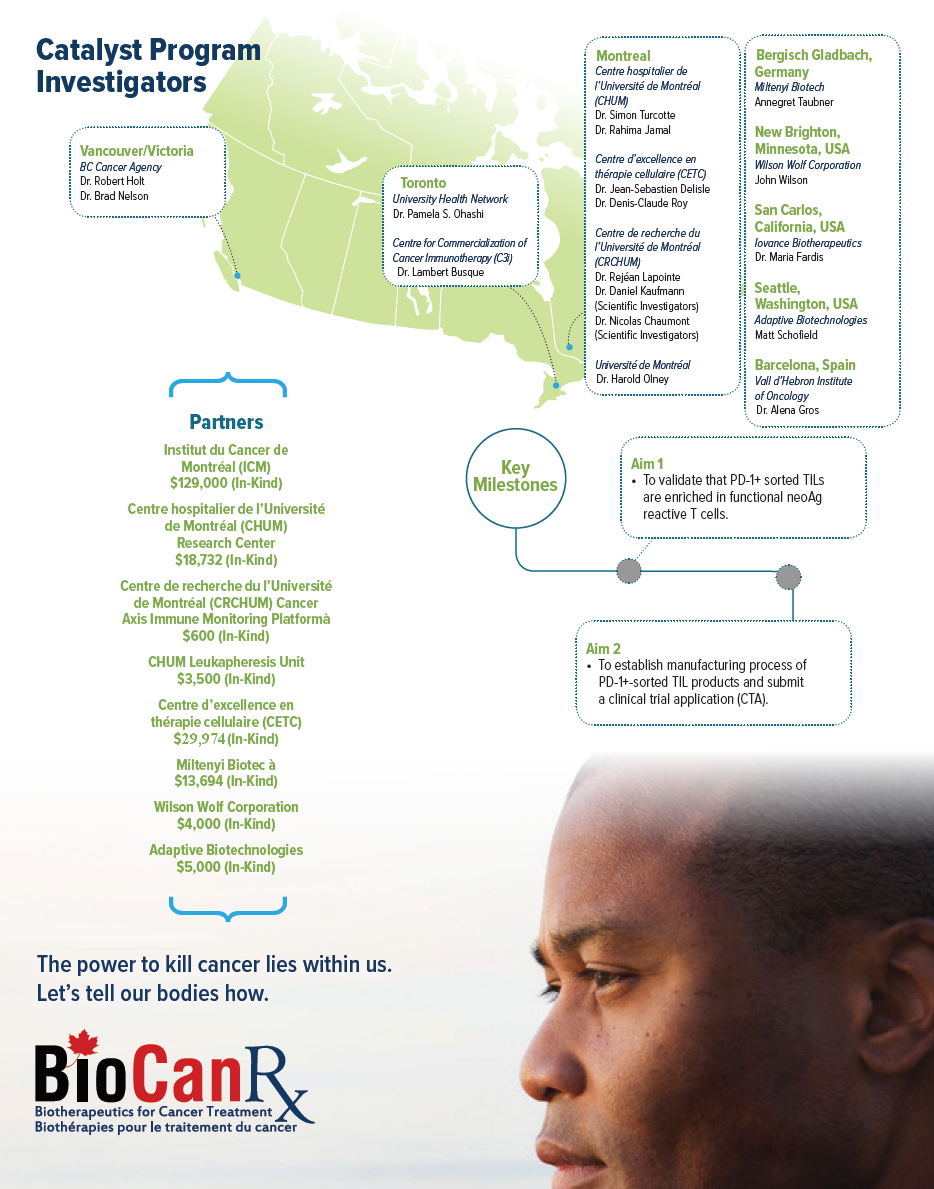
Project summary: Catalyst Program
Improving adoptive cell transfer immunotherapy with clinical-grade cell sorting of tumour-reactive T-cells infiltrating solid tumours
April 23rd, 2018 to March 31st, 2021
HIGHLIGHTS

- This project will generate scientific evidence showing the superior features of the tumourreactive T-cell product in comparison to one obtained with conventional tumour-infiltrating T-cell manufacturing
- The results of the proposed study will be utilized by other BioCanRx network investigators planning to use cell sorting to improve the efficacy of other types of adoptive cell transfer immunotherapy
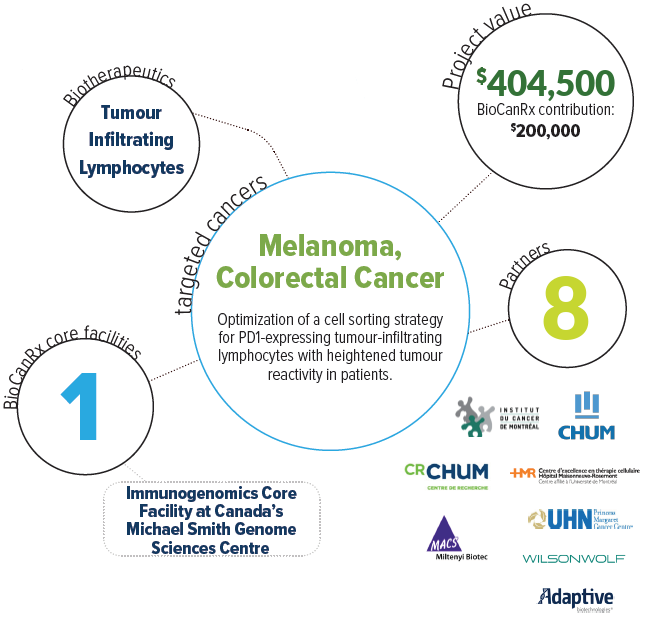
About the Project
Cancer immunotherapy, which uses the immune system to destroy cancer cells, is a real medical breakthrough. However, current strategies mainly rely on causing a general immune boost, which benefits only a minority of patients with tumours well recognized by the immune system. A small number of anti-tumour immune cells, called T-cells, can naturally infiltrate tumours in most patients. One powerful approach to cancer immunotherapy is to make a cell transfusion product out of tumour-infiltrating T-cells produced in large numbers outside the body. We propose to enhance this approach, called adoptive cell transfer immunotherapy, by making a cell transfusion product highly enriched in tumour-reactive T-cells. This will be achieved by selecting tumour-infiltrating T-cells expressing a marker called PD-1, that acts as a “tag” for tumour-reactive T-cells. To do that, we have access to a sophisticated new device called a cell sorter, currently unique in Canada. The main goal of this project is to get the approval from Health Canada to use the tumour-reactive T-cell product in a clinical trial within two years. We will also generate scientific evidence showing the superior features of this cell product in comparison to one obtained with conventional tumour-infiltrating T-cell manufacturing. The knowledge generated throughout the project will serve other BioCanRx network investigators planning to use cell sorting to improve the efficacy of other types of adoptive cell transfer immunotherapy.