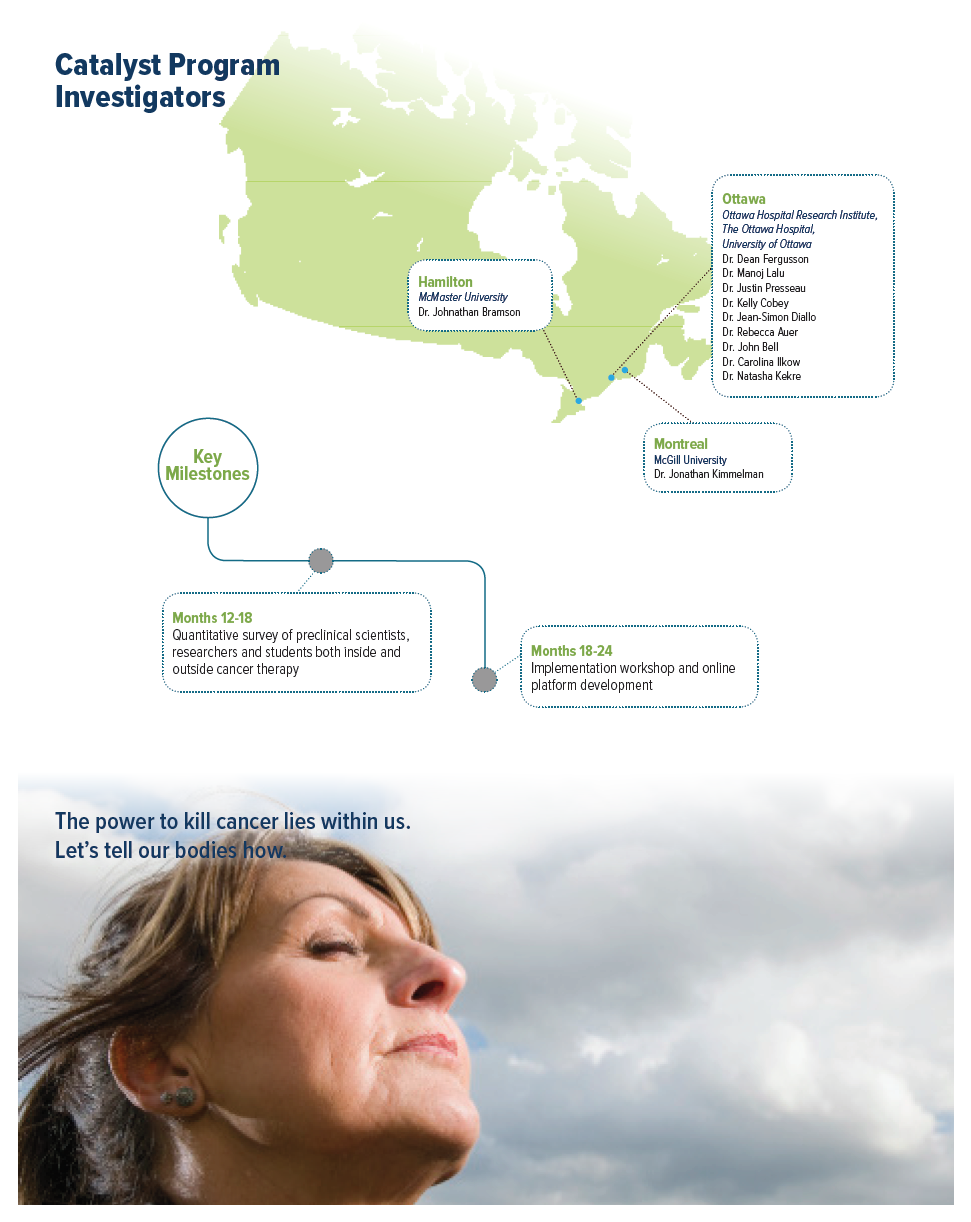
Project summary: Clinical, Social and Economic Impact Program
Translational Cancer Immunotherapeutics: Improving the Design, Analysis, and Reporting of Preclinical Studies
Jan 17, 2017 to Dec 31, 2019
HIGHLIGHTS

- First empirical approach using state of the art methods to assess experimental design and reporting in preclinical cancer biotherapeutics
- Responds to the reports of irreproducible preclinical research
- Maximizes the number of promising therapeutics successfully translated to clinical trials by improving the quality of experimental design and reporting by BioCanRx investigators

About this project
Experiments which are poorly designed and poorly reported waste valuable time and resources. Poor reporting and design in preclinical animal research contributes to the ‘replication crisis’ that has been recently highlighted by funders and high level journals. Moreover, suboptimal reporting and design have contributed to flawed decisions to proceed to first-in-human clinical trials that ultimately failed.
In response to reports of irreproducible preclinical research, funders and journals are enforcing new guidelines on how preclinical research should be conducted and reported. Applying the correct design and analysis of experiments lessens bias and increases the likelihood of identifying cause-effect relationships between therapies and disease response. Transparent reporting ensures that the end users can evaluate the strength of the evidence for claims of cause-effect relationships. These are critical components to select therapies that are most likely to benefit patients. Currently, very little is known about the quality of experimental design and reporting in in vivo preclinical cancer therapy research let alone in cancer immunotherapy.
If there is room for improvement, cancer scientists need to be engaged to understand their culture, attitudes and needs, as well as the barriers and facilitators, for implementing mechanisms to improve experimental design and reporting. To address these gaps the team proposes to conduct a scoping review to better understand current norms of design, analysis, and reporting of in vivo preclinical cancer immunotherapy studies; to conduct a mixed methods knowledge translation project to assess the attitudes, barriers, and facilitators to implementing changes in experimental design and reporting; and based on findings from the first two, to develop a comprehensive workshop and online resources on in vivo preclinical research design and reporting.
Ensuring that scientists and trainees are implementing rigorous experimental designs and clearly reporting their findings will maximise the number of promising therapeutics successfully translated to clinical trials. This project will be the first empirical approach, using state of the art methods in scoping reviews and knowledge translation, to assess experimental design and reporting in preclinical cancer biotherapeutics. This team of researchers led by Dr. Fergusson will create an evidence map that will explicitly describe the current state of design, reporting, and analysis in cancer biotherapeutics.
Importantly, the map will identify areas for potential improvement while benchmarking the current state of research. This project will directly engage preclinical cancer scientists ensuring that their experience and expertise is used to identify current understanding of preclinical reporting and experimental design. This will identify barriers and facilitators to implement best practices for reporting and design.