Supporting Home Grown Innovation
Challenges
Building Canadian Capacity
The Canadian scientific community is consistently recognized internationally for its excellence in health research.
However, without affordable access to the expertise and infrastructure required to translate discoveries into innovative therapies that are safe to be tested in the clinic, this world-class research will either:
- Continue to languish in the laboratory
- Travel beyond Canada’s borders to realize its full potential.
The Opportunity
The Need for Biomanufacturing
The advent of disruptive biologic technologies has ushered in an unprecedented need for the highly specialized field of biotherapeutics manufacturing (biomanufacturing).
Currently, there are significant gaps in Canada’s ability to produce lifesaving, biologic-based medicines such as vaccines and therapeutic drugs at a sufficient scale to meet both domestic and global demand.
Establishing domestic biomanufacturing capacity in Canada is critical to building a pipeline of novel therapies for the benefit of Canadian patients, and is integral to anchoring a robust, burgeoning innovation economy in the Canadian life science sector.

BioCanRx’s Response
- Bridging the clinical translation gap
- Building Canadian GMP biomanufacturing capacity
- Expanding access to critical translational biomanufacturing assets
- Bringing new biomanufacturing capacity online for novel therapies like CAR T
Approach
Expanding Through a Network Approach
The growing demand, in tandem with the increased number of approvals for new disruptive biologically-based medicines, led BioCanRx to expand its initial investment to apply a network approach to biomanufacturing.
This biomanufacturing network:
- Links existing Canadian facilities and expertise
- Harmonizes standard operating procedures and best practices
- Supports unprecedented demand for high-quality biologically-based medicines
Through targeted investments in knowledge translation and training, the network will:

Challenges
Expanding and Optimizing Canada’s GMP Vector Manufacturing Capacity
The global demand for access to biotherapeutics manufacturing was already critical pre-COVID-19—and the pandemic only magnified this need. Wait times for clinical drug product manufacturing often exceeded 18 months. Reliance on costly international organizations limited the number of therapies that could be tested in Canada.
Without intervention, Canadian innovation risked:
- Long delays in clinical evaluation
- Barriers to developing homegrown therapies
The Opportunity
BioCanRx Investments
To address this Canadian biomanufacturing gap, BioCanRx made targeted investments to support end-to-end GMP manufacture of virus-based therapeutics, inclusive of fill finish, to drive the evaluation of novel products in Canada. Leveraging investments made in both Cycle 1 and 2, BioCanRx is encouraging long-term sustainable growth within Canada’s biomanufacturing sector by providing operational know-how, knowledge translation and training support to two of Canada’s existing biomanufacturing facilities.

Lasting Impact
- Next-generation cancer immunotherapies
- Therapies for rare diseases
Closing Canada’s Skills Gap
Cultivating Biotherapeutics Expertise
BioCanRx has put several strategic initiatives in place to create an academic to industry pipeline that addresses the significant skills gap in biomanufacturing that exists in Canada today.
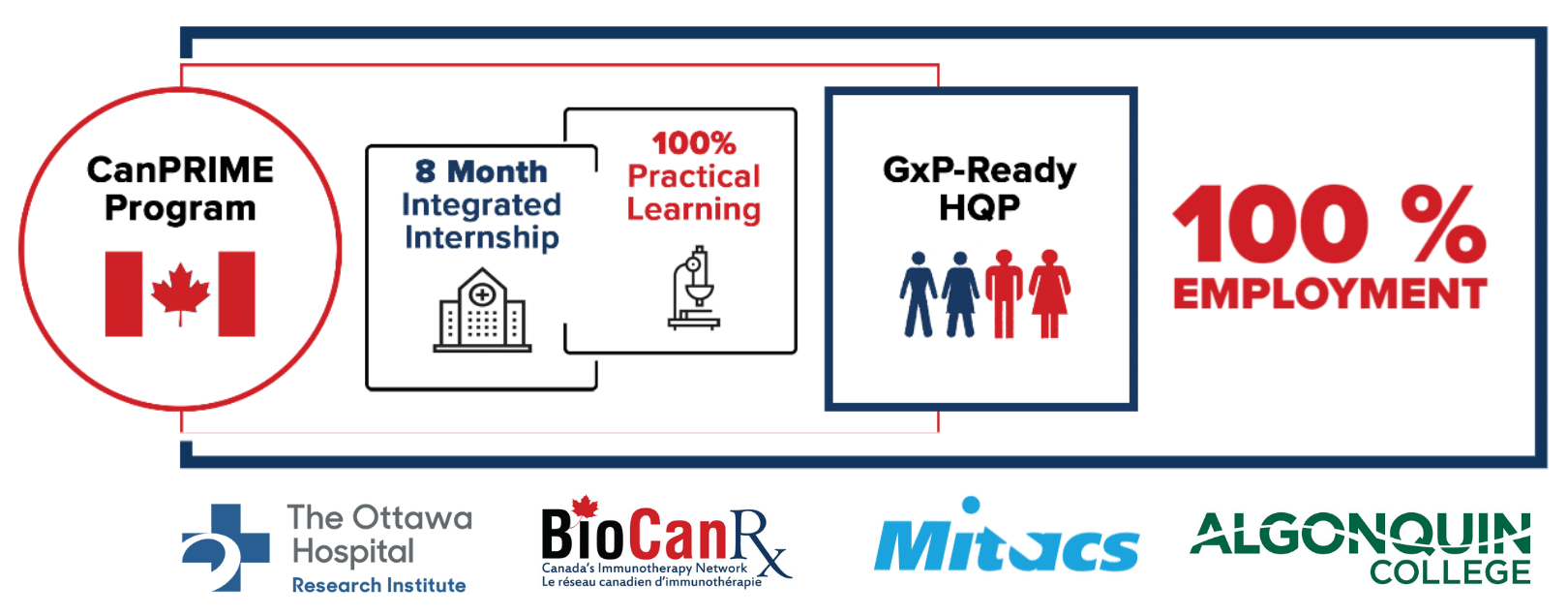
The Start: CanPRIME
In 2018, The Ottawa Hospital Research Institute’s Biotherapeutics Manufacturing Centre (BMC), Algonquin College, the University of Ottawa, and Mitacs launched CanPRIME, a first-of-its-kind Good Manufacturing Practices (GMP) training program. Students gained hands-on experience in GMP biomanufacturing.
National Expansion: CanPRIME 2.0
By 2022, BioCanRx and partners expanded the program nationally, adding five new GMP training sites across:
- British Columbia
- Alberta
- Manitoba
- Saskatchewan
- Ontario












8-Month Paid Internship
Work-integrated training at GMP facilities.
Hands-On GMP Learning
Practical application of GMP principles.
Standardized Curriculum
Addressing labour market needs across all training nodes.
Hybrid Training
Both on-the-job and off-the-job experiences.
Extra Support
Funding for external training, lab exchanges, and an online GxP microcredential (Algonquin + BioCanRx).
CanPRIME 2.0 will support the onboarding and training of more than 70 trainees from university and college at BioCanRx’s training nodes, creating a pipeline of HQP armed with invaluable experience and marketability for future employment opportunities within the biomanufacturing sector. Employability of CanPRIME ‘graduate’ interns currently sits at 100%, demonstrating both the need and high employability of these GMP-ready individuals.
CanPRIME and its national expansion, CanPRIME 2.0, represent a unique and novel approach to provide experienced, job-ready individuals to provision industry, academia and not-for-profits with GMP advanced therapeutic product biomanufacturing know-how.

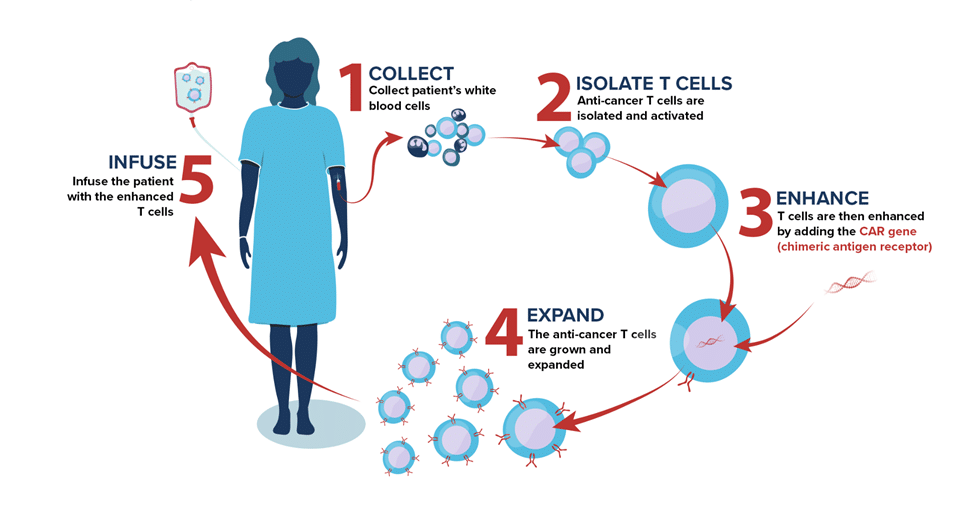
Despite ongoing success treating previously untreatable disease with CAR T-cell therapy, access to this transformative treatment is largely missing in Canada. As with many personalized therapies, CAR T carries with it a significant up-front manufacturing cost and the use of GMP biomanufacturing facilities. To address this, BioCanRx developed a network of “point-of-care” (POC) manufacturing facilities across the country that provide greater access to innovative adoptive cell therapies (ACT) like CAR T for Canadian patients.